|
Magazine issue 2827 from NewScientist Website
Electric ice may pervade space.
This strange form of water is more
persistent than was previously thought, and the discovery could
change our understanding of how the solar system formed. It might
even give ice a new role in the emergence of the
complex organic
molecules needed for life.
Icy planet rings might be highly charged (Image: JPL/NASA)
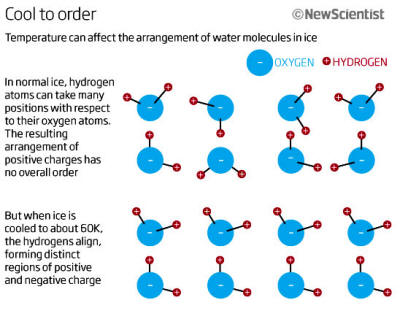
That's because the two positively charged hydrogen atoms cluster at one end, away from the single negatively charged oxygen. However, the charges get mixed up when ordinary ice, known as Ice Ih, forms.
While the oxygen atoms arrange themselves in a repeating pattern, the pairs of hydrogen atoms that extend from them don't.
Instead, they randomly take one of a number of positions (see
below graphic).
Cool ice to about 60 kelvin (-213.15 °C), though, and the hydrogens
rearrange themselves so they are aligned. In the resulting,
perfectly regular crystal, called
Ice XI, there are distinct regions
of positive and negative charge.
Electric ice could also
attract organic compounds, possibly accelerating the
emergence of
complex molecules and eventually life.
They haven't directly detected it there,
however.
In 2006,
Masashi Arakawa, now at Kyushu University in
Fukuoka, Japan, and colleagues produced Ice XI in the lab between
57K and 66K. This is about the temperature found on Uranus and its
moons, but too narrow a range for Ice XI to be a major player in
planet formation.
Tiny flecks of the stuff embedded in
ordinary ice can help convert all of it to electric ice, a little
like the ice dreamed up by Kurt Vonnegut in his novel Cat's Cradle,
a single crystal of which could freeze the world's oceans.
Crucially, the conversion of Ice Ih to Ice XI occurs at
higher temperatures than if Ice XI forms from scratch.
Then they warmed it to 100 K so that it
reverted to ordinary ice. When they turned the thermostat back down,
Ice XI returned at a higher temperature than before: 72 K. Yet
cooling regular ice to 72 K did not change its structure
(Geophysical Research Letters,
DOI: 10.1029/2011GL048217).
Further experiments revealed that this
memory effect happens even when Ice XI is heated to 111 K, the sort
of temperature found on the moons of Jupiter. That suggests Ice XI
may be far more common in space than thought.
That is relatively rare in space but if they can demonstrate that some more common substance, such as ammonia or methane, can also catalyze ice XI's formation,
|