28 July, 2012
from
RT Website
Pentagon’s cutting
edge research lab says that
they’ve used a massive
harvest of tobacco plants
to help produce a plethora of
flu-fighting vaccines.
The Pentagon’s
DARPA lab has announced a milestone, but it
doesn’t involve drones or death missiles.
Scientists at the Defense Advanced Research
Projects Agency say they’ve produced 10 million doses of an influenza
vaccine in only one month’s time. In a press release out of the agency’s
office this week, scientists with DARPA say they’ve reach an important step
in being able to combat a flu pandemic that might someday decimate the
Earth’s population.
By working with the
Medicago Inc. vaccine company, the
Pentagon’s cutting edge research lab says that they’ve used a massive
harvest of tobacco plants to help produce a plethora of flu-fighting
vaccines.
“Testing confirmed that a single dose of the
H1N1 VLP influenza vaccine candidate induced protective levels of
hemagglutinin antibodies in an animal model when combined with a
standard aluminum
adjuvant,” the agency writes, while
still noting, though, that “the equivalent dose required to protect
humans from natural disease can only be determined by future,
prospective clinical trials.”
Researchers have before relied on using chicken
eggs to harvest compounds to use in influenza vaccines.
With a future outbreak requiring scientists to
step up with a solution as soon as possible, though, they’ve turned to
tobacco plants to help produce the vaccines.
“Vaccinating susceptible populations during
the initial stage of a pandemic is critical to containment,” Dr. Alan
Magill, DARPA program manager, says in an official statement.
“We’re looking at plant-based solutions to
vaccine production as a more rapid and efficient alternative to the
standard egg-based technologies, and the research is very promising.”
The World Health Organization has gone on
the record to say that as much as half of the people on the planet could be
affected by a pandemic in the near future, and it could take as much as nine
months for a vaccine for a pandemic virus strain to become made available.
With the lives of billions of people across the
world at stake, DARPA has been trying to determine new ways of churning out
antidotes in as little time as possible.
Now its researchers say, that in only a month,
scientists,
“produced more than 10 million doses (as
defined in an animal model) of an H1N1 influenza vaccine candidate based
on virus-like particles (VLP).”
Through DARPA’s previously established Blue
Angel program (below insert), researchers have spent several years
searching for new ways to produce mass quantities of vaccine-grade protein
that could be used to combat what they say are very real emerging and novel
biological threats.
|
- H1N1 Acceleration
-
Blue Angel
from
DARPA Website
In May 2009, DARPA initiated the Blue Angel effort to identify
ongoing programs to assist in the Government-wide response to
the H1N1 pandemic.
The Blue Angel program is an accelerated
and integrated effort to deliver effective interventions for
pandemic influenza.
Blue Angel brings together the following
technologies to form a comprehensive approach in response to a
pandemic influenza or manmade outbreak:
-
Predicting Health and Disease (PHD),
a program to predict and diagnose individuals exposed to
influenza before they are symptomatic
-
Modular IMmune In vitro Constructs
(MIMIC®), a program to identify safe and effective treatments in
a test tube
-
Accelerated Manufacture of
Pharmaceuticals (AMP), a capability for rapidly mass producing
low-cost, vaccine-grade recombinant protein that has the
potential for scale up to tens of millions of doses per month
In response to the 2009 H1N1 swine flu
pandemic, Blue Angel programs are currently in a "live-fire test" to
demonstrate a flexible and agile capability for the Defense
Department to rapidly react and neutralize any natural or
intentional pandemic disease.
-
Predicting Health and Disease (PHD):
PHD has developed a method for
determining who will or will not become sick after exposure to a
virus many days before symptoms appear, typically within 10
percent of the incubation period of a particular virus. This is
accomplished using a highly accurate, mRNA-based blood test.
By identifying key biomarkers for
host response to respiratory viral infections, PHD can
categorize viral-exposed individuals into specific categories -
those who will be sick, those individuals who are contagious,
and those who are well.
Accuracy of this method is 85-90
percent within hours of viral exposure and achieves near
100-percent detection after a few days. High accuracy of
detection enables prevention, prediction of disease propagation,
and appropriate early treatment of infected individuals.
-
Accelerated Manufacture of
Pharmaceuticals (AMP):
This program seeks to identify new
ways to produce large amounts of high-quality vaccine-grade
protein in less than 3 months in response to emerging and novel
biologic threats.
In response to the 2009 H1N1 swine
flu pandemic, as a "live-fire test," the plant platform
redirected its rapid scale-up process developed for avian
influenza to the new H1N1 virus and produced a recombinant
protein within 4 weeks.
-
Modular IMmune In vitro Constructs
(MIMIC®):
As animal studies are not always a
good predictor of a vaccine's safety and efficacy in a human,
the MIMIC® system will work in parallel with the AMP program to
test the subunit of vaccine produced under the AMP program to
ensure it is safe and immunogenic.
|
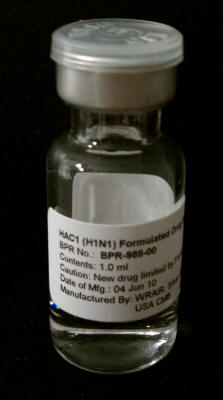
Andy Sheldon, Chief Executive Officer of Medicago , says in the
company’s own press release that
"The completion of the rapid fire test marks
a substantial achievement in demonstrating our technology and the
potential for Medicago to be the first responder in the event of a
pandemic flu outbreak.”
Medicago’s research was conducted in a
97,000-square-foot vaccine facility in North Carolina that was funded
through a $21 million Technology Investment Agreement with DARPA.