|
by Shannon Brownlee
and Jeanne Lenzer from TheAtlantic Website
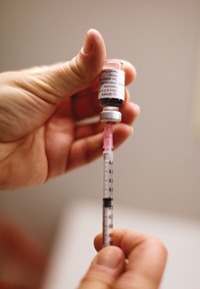
Drive too fast along Red Lion Road, beside Philadelphia’s Northeast Airport, and you will miss the low-rise cement building where the biotech company MedImmune has been quietly pumping out swine flu vaccine at about a million doses a week.
Through the summer and fall, workers wearing protective gear that covered them from head to toe brewed up batches of live, genetically modified flu virus. Robots then injected tiny doses of virus-laden fluid into glass vials, which were mounted into nasal spritzers, labeled, and readied for shipment at the direction of the Centers for Disease Control and Prevention, in Atlanta, which is helping to coordinate the nation’s pandemic-preparedness plan.
In
the most ambitious vaccination program the nation has mounted since
the anti-polio campaign in the 1950s, the federal government has
commissioned Med-Immune and four other companies to produce enough
vaccine to cover the entire U.S. population.
In August, New
York state required all health-care workers to get both seasonal and
swine flu shots. To further protect the populace, the federal
government has spent upwards of $3billion stockpiling millions of
doses of antiviral drugs like Tamiflu—which are being used both to
prevent swine flu and to treat those who fall ill.
The U.S. government—with the support of leaders in the public-health and medical communities—has put its faith in the power of vaccines and antiviral drugs to limit the spread and lethality of swine flu. Other plans to contain the pandemic seem anemic by comparison. Yet some top flu researchers are deeply skeptical of both flu vaccines and antivirals.
Like the engineers who warned for
years about the levees of New Orleans, these experts caution that
our defenses may be flawed, and quite possibly useless against a
truly lethal flu. And that unless we are willing to ask fundamental
questions about the science behind flu vaccines and antiviral drugs,
we could find ourselves, in a bad epidemic, as helpless as the
citizens of New Orleans during Hurricane Katrina.
More than 200 known viruses and
other pathogens can cause the suite of symptoms known as
“influenza-like illness”; respiratory syncytial virus, bocavirus,
coronavirus, and rhinovirus are just a few of the bugs that can make
a person feel rotten. And depending on the season, in up to
two-thirds of the cases of flu-like illness, no cause at all can be
found.
What is certain is that influenza viruses mutate with amazing speed, so each flu season sees slightly different genetic versions of the viruses that infected people the year before.
Every year, the World Health Organization and the Centers
for Disease Control and Prevention collect data from 94 nations on
the flu viruses that circulated the previous year, and then make an
educated guess about which viruses are likely to circulate in the
coming fall. Based on that information, the U.S. Food and Drug
Administration issues orders to manufacturers in February for a
vaccine that includes the three most likely strains.
It is these novel viruses that are responsible for pandemics, defined by the World Health Organization as events that occur when “a new influenza virus appears against which the human population has no immunity” and which can sweep around the world in a very short time. The worst flu pandemic in recorded history was the “Spanish flu” of 1918–19, at the end of World WarI.
A third of the world’s population
was infected, with at least 40million and perhaps as many as
100million people dying—more than were killed in World Wars I and II
combined. (Some scholars suggest that one reason World War I ended
was that so many soldiers were sick or dying from flu.) Since then,
two other flu pandemics have occurred, in 1957 and 1968, neither of
which was particularly lethal.
For example, Australian officials estimated
they would finish winter with under 1,000 swine flu deaths—fewer
than the usual 1,500 to 3,000 from seasonal flu. Among those who
have died in the U.S., about 70 percent were already suffering from
congenital conditions like cerebral palsy or underlying illnesses
such as cancer, asthma, or AIDS, which make people more vulnerable.
Influenza virus was first identified in the 1930s, and by the mid-1940s, researchers had produced a vaccine that was given to soldiers in World War II. The U.S. government got serious about promoting flu vaccine after the 1957 flu pandemic brought home influenza’s continuing potential to cause widespread illness and death.
Today, flu vaccine is a staple
of public-health policy; in a normal year, some 100 million
Americans get vaccinated.
For this reason,
researchers studying the impact of flu vaccination typically look at
deaths from all causes during flu season, and compare the vaccinated
and unvaccinated populations.
So how could flu vaccine possibly reduce total deaths by half?
Tom Jefferson, a physician based in Rome and the head of the Vaccines Field at the Cochrane Collaboration, a highly respected international network of researchers who appraise medical evidence, says:
The estimate of 50 percent mortality reduction is based on “cohort studies,” which compare death rates in large groups, or cohorts, of people who choose to be vaccinated, against death rates in groups who don’t. But people who choose to be vaccinated may differ in many important respects from people who go unvaccinated—and those differences can influence the chance of death during flu season.
Education, lifestyle, income, and many other “confounding” factors can come into play, and as a result, cohort studies are notoriously prone to bias.
When researchers crunch the numbers, they typically try to factor out variables that could bias the results, but, as Jefferson remarks,
There is always the chance that they might
miss some critical confounder that renders their results entirely
wrong.
They hypothesized that on average, people who get vaccinated are simply healthier than those who don’t, and thus less liable to die over the short term. People who don’t get vaccinated may be bedridden or otherwise too sick to go get a shot. They may also be more likely to succumb to flu or any other illness, because they are generally older and sicker.
To test their thesis,
Jackson and her colleagues combed through eight years of medical
data on more than 72,000 people 65 and older. They looked at who got
flu shots and who didn’t. Then they examined which group’s members
were more likely to die of any cause when it was not flu season.
In fact, the healthy-user effect explained the entire benefit that other researchers were attributing to flu vaccine, suggesting that the vaccine itself might not reduce mortality at all.
Jackson’s papers “are beautiful,” says Lone Simonsen, who is a professor of global health at George Washington University, in Washington, D.C., and an internationally recognized expert in influenza and vaccine epidemiology.
The results were also so unexpected that many experts simply refused to believe them. Jackson’s papers were turned down for publication in the top-ranked medical journals.
One flu expert who reviewed her studies for the Journal of the American Medical Association wrote,
When the papers were finally published in 2006, in the less prominent International Journal of Epidemiology, they were largely ignored by doctors and public-health officials.
The history of flu vaccination suggests other reasons to doubt claims that it dramatically reduces mortality.
In 2004, for example, vaccine production fell behind, causing a 40 percent drop in immunization rates. Yet mortality did not rise. In addition, vaccine “mismatches” occurred in 1968 and 1997: in both years, the vaccine that had been produced in the summer protected against one set of viruses, but come winter, a different set was circulating. In effect, nobody was vaccinated.
Yet death rates from all causes, including flu and the various illnesses it can exacerbate, did not budge.
Sumit Majumdar, a physician and researcher at the University
of Alberta, in Canada, offers another historical observation: rising
rates of vaccination of the elderly over the past two decades have
not coincided with a lower overall mortality rate. In 1989, only 15
percent of people over age 65 in the U.S. and Canada were vaccinated
against flu. Today, more than 65 percent are immunized. Yet death
rates among the elderly during flu season have increased rather than
decreased.
The proponents suggest, for instance, that
influenza viruses may have become more contagious over time, and
thus are infecting greater numbers of elderly people, including some
who have been vaccinated. Or maybe the viruses are becoming more
lethal. Or maybe the elderly have less immunity to flu than they
once did because, say, their diets have changed.
Nancy Cox, the CDC’s influenza division chief, says flatly,
Anthony Fauci, a physician and the director of the National Institute of Allergy and Infectious Diseases at the NIH, where much of the basic science of flu vaccine has been worked out, says,
Majumdar says,
The most vocal—and undoubtedly most vexing—critic of the gospel of flu vaccine is the Cochrane Collaboration’s Jefferson, who’s also an epidemiologist trained at the famed London School of Tropical Hygiene, and who, in Lisa Jackson’s view, makes other skeptics seem “moderate by comparison.”
Among his fellow flu researchers, Jefferson’s outspokenness has made him something of a pariah. At a 2007 meeting on pandemic preparedness at a hotel in Bethesda, Maryland, Jefferson, who’d been invited to speak at the conference, was not greeted by any of the colleagues milling about the lobby. He ate his meals in the hotel restaurant alone, surrounded by scientists chatting amiably at other tables. He shrugs off such treatment.
As a medical officer working for the United Nations in 1992, during the siege of Sarajevo, he and other peacekeepers were captured and held for more than a month by militiamen brandishing AK-47s and reeking of alcohol.
Professional shunning seems trivial by comparison, he says.
Yet while other flu researchers may not like what Jefferson has to say, they cannot ignore the fact that he knows the flu-vaccine literature better than anyone else on the planet.
He leads an international team of researchers who have combed through hundreds of flu-vaccine studies. The vast majority of the studies were deeply flawed, says Jefferson.
Only four studies were properly designed to pin down the effectiveness of flu vaccine, he says, and two of those showed that it might be effective in certain groups of patients, such as school-age children with no underlying health issues like asthma.
The other two showed equivocal
results or no benefit.
Likewise, nobody really has a handle on how many of the deaths that are blamed on flu were actually caused by a flu virus, because few are confirmed by a laboratory.
There’s one other way flu researchers may be fooled into thinking flu vaccine is effective, Jefferson says.
All vaccines work by
delivering a dose of killed or weakened virus or bacteria, which
provokes the immune system into producing antibodies. When the
person is subsequently exposed to the real thing, the body is
already prepared to repel the bug completely or to get rid of it
after a mild illness. Flu researchers often use antibody response as
a way of gauging the effectiveness of vaccine, on the assumption
that levels of antibodies in the blood of people who have been
vaccinated are a good predictor—although an imperfect one—of how
well they can ward off the infection.
Studies show that young, healthy people mount a glorious immune response to seasonal flu vaccine, and their response reduces their chances of getting the flu and may lessen the severity of symptoms if they do get it.
But they aren’t the people who die from seasonal flu. By contrast, the elderly, particularly those over age70, don’t have a good immune response to vaccine—and they’re the ones who account for most flu deaths.
(Infants with severe disabilities, such as leukemia and
congenital lung disease, and people who are immune-compromised—from
AIDS, or diabetes, or cancer treatment—make up the rest. As of
August8, only 36 deaths from swine flu had been confirmed among
children in the U.S., and the overwhelming majority of those
children had multiple, severe health disorders.)
These
questions have led to the most controversial aspect of Jefferson’s
work: his call for placebo-controlled trials, studies that would
randomly give half the test subjects vaccine and the other half a
dummy shot, or placebo. Only such large, well-constructed,
randomized trials can show with any precision how effective vaccine
really is, and for whom.
Lone Simonsen explains the prevailing view:
They feel strongly that vaccine has been shown to be effective and that a sham vaccine would put test subjects at unnecessary risk of getting a serious case of the flu.
In a phone interview, Fauci at first voiced the opinion that a placebo trial in the elderly might be acceptable, but he called back later to retract his comment, saying that such a trial “would be unethical.”
Jefferson finds this view almost exactly backward:
Just after 6 p.m. on a warm Friday evening in July, Dr. David Newman is only minutes into a 10-hour shift in the emergency room of New York City’s St. Luke’s Hospital, and already he has assumed responsibility for 11 patients.
The young Italian tourist sitting on the bed in front of the doctor has meningitis, and through an interpreter, Newman tells him he almost certainly has the viral form of the disease, which will do nothing more than make him feel ill for a few days. There is a tiny chance, says Newman, that the illness is caused by a bacterium, which can be deadly, but he is almost positive that’s not what the tourist has.
He says to his patient,
The tourist, on learning that he might be infected with a potentially lethal disease, looks down at his feet and confesses that he is much more worried about another illness: swine flu.
Newman smiles patiently.
Late last spring, as headlines and airwaves warned of a possible pandemic, patients like Newman’s began clogging emergency rooms across the country, a sneezing, coughing, infectious tide of humanity more worried than truly sick, but whose mere presence in the emergency room has endangered the lives of others.
In an average year the ER at St. Luke’s, a sprawling 1,076-bed hospital on 113th Street, takes in 110,000 patients, some 300 a day.
At the height of the summer swine flu outbreak, that
number doubled. The vast majority of panicky patients who came in
the door at St. Luke’s and other emergency departments didn’t
actually have the virus, and of those who did, most were not sick
enough to need hospitalization. Even so, says Newman, when patients
with even mild flu symptoms show up in the hospital, they vastly
increase the spread of the virus, simply because they inevitably
sneeze and cough in rooms that are jammed with other people.
Public-health officials
at the state and local levels are also recommending the drugs.
Guidelines issued by the New York City Department of Health, says
Newman, “encourage us to give a prescription to just about every
patient with the sniffles,” a practice that some experts worry will
quickly lead to resistant strains of the virus.
Daniel Janies, an associate professor of biomedical informatics at Ohio State University, tracks the genetic mutations that allow flu virus to develop resistance to drugs. Flu can become resistant to Tamiflu in a matter of days, he says. Handing out the drug early in the pandemic, when H1N1 poses only a minimal threat to the vast majority of patients, strikes him as “shortsighted.”
Indeed, samples of resistant H1N1 were cropping up by midsummer, increasing the likelihood that come late fall, many people will be infected with a resistant strain of swine flu. Alarmed at that prospect, the World Health Organization issued an alert on August 21, recommending that Tamiflu and Relenza be used only in severe cases and in patients who are at high risk of serious complications.
By mid-August, two U.S. swine flu patients had
developed Tamiflu-resistant strains.
This was after Congress had already approved $1.8billion to stockpile Tamiflu for the military, a decision that was made during the tenure of Defense Secretary Donald Rumsfeld.
(Before joining the Bush Cabinet, Rumsfeld was chairman
for four years of Gilead Sciences, the company that holds the patent
on Tamiflu, and he held millions of dollars’ worth of stock in the
company. According to Roll Call, an online newspaper covering events
on Capitol Hill, Rumsfeld says he recused himself from all
government decisions involving Tamiflu. Gilead’s stock price rose
more than 50 percent in 2005, when the government’s plan was
announced.)
On average, Tamiflu (which accounts for 85 to 90 percent of the flu antiviral-drug market) cuts the duration of flu symptoms by 24hours in otherwise healthy people. In exchange for a slightly shorter bout of illness, as many as one in five people taking Tamiflu will experience nausea and vomiting. About one in five children will have neuropsychiatric side effects, possibly including anxiety and suicidal behavior.
In Japan, where Tamiflu is liberally prescribed, the drug may have been responsible
for 50 deaths from cardiopulmonary arrest, from 2001 to 2007,
according to Rokuro Hama, the chair of the Japan Institute of
Pharmacovigilance.
Roche Laboratories, the company licensed to manufacture and market Tamiflu, says its drug does just that. In two September 2006 press releases, the company announced,
Once again cohort studies (the same kind of potentially biased research that led to the conclusion that flu vaccine cuts mortality by 50 percent) are behind these claims.
Tamiflu costs $10 a pill. It is possible
that people who take it are more likely to be insured and affluent,
or at least middle-class, than those who do not, and a large body of
evidence shows that the well-off nearly always fare better than the
poor when stricken with an infectious disease, including flu. In
both 2003 and 2009, reviews of randomized placebo-controlled studies
found that the study populations simply weren’t large enough to
answer the question: Does Tamiflu prevent pneumonia?
An FDA
spokesperson said recently that the agency is unaware of any data
submitted by Roche that would support the claims in the company’s
September 2006 news release about the drug’s reducing flu deaths.
The short answer may be that public-health officials feel they must offer something, and these drugs are the only possible remedies at hand.
The CDC’s Nancy Cox also acknowledges that the science is not
as sound as she might like, but the government still recommends
their use. And as with vaccines, she considers additional randomized
placebo-controlled trials of the antiviral drugs to be “unethical”
and thus out of the question.
When faced with contrary evidence, the adherents acknowledge that the science is not as crisp as they might wish.
Then, in response to calls for placebo-controlled trials, which would provide clear results one way or the other, the proponents say such studies would deprive patients of vaccines and drugs that have already been deemed effective.
Students of U.S. medical history will find this circular logic familiar: it is a long-recurring theme in American medicine, and one that has, on occasion, had deadly consequences.
In 1925, Sinclair Lewis caricatured a medical culture that allowed belief—and profits—to distort science in his Pulitzer Prize–winning book, Arrowsmith. Based on the lives of the real-life microbiologists Paul de Kruif and Jacques Loeb, Lewis tells the story of Martin Arrowsmith, a physician who invents a new vaccine during a deadly outbreak of bubonic plague.
But his efforts to test the vaccine’s
efficacy are frustrated by an angry community that desperately wants
to believe the vaccine works, and a profit-hungry institute that
rushes the vaccine into use prematurely—forever preempting the
proper studies that are needed.
In the 1980s and ’90s, for example, cancer specialists were convinced that high-dose chemotherapy followed by a bone-marrow transplant was the best hope for women with advanced breast cancer, and many refused to enroll their patients in randomized clinical trials that were designed to test transplants against the standard—and far less toxic—therapy. The trials, they said, were unethical, because they knew transplants worked. When the studies were concluded, in 1999 and 2000, it turned out that bone-marrow transplants were killing patients.
Another recent example involves drugs related to the analgesic lidocaine. In the 1970s, doctors noticed that the drugs seemed to make the heart beat rhythmically, and they began prescribing them to patients suffering from irregular heartbeats, assuming that restoring a proper rhythm would reduce the patient’s risk of dying.
Prominent cardiologists for years opposed clinical
trials of the drugs, saying it would be medical malpractice to
withhold them from patients in a control group. The drugs were
widely used for two decades, until a government-sponsored study
showed in 1989 that patients who were prescribed the medicine were
three and a half times as likely to die as those given a placebo.
It might sound coldhearted, but it is the only way to know for certain whether, and for whom, current remedies actually work. It would also be useful to know whether vaccinating healthy people—who can mount an immune response on their own—protects the more vulnerable people around them. For example, immunizing nursing-home staff and healthy children is thought to reduce the spread of flu to the elderly and the immune-compromised.
Pinning down the effectiveness of this strategy
would be a bit more complex, but not impossible.
As it stands, more than 50 percent of health-care workers say they do not intend to get vaccinated for swine flu and don’t routinely get their shots for seasonal flu, in part because many of them doubt the vaccines’ efficacy. The other possibility, of course, is that we’re relying heavily on vaccines and antivirals that simply don’t work, or don’t work as well as we believe.
And as a result, we may be neglecting other, proven measures that could minimize the death rate during pandemics.
For instance, health departments in every state are responsible for submitting plans to the CDC for educating the public, in the event of a serious pandemic, about hand-washing and “social distancing” (voluntary quarantines, school closings, and even enforcement of mandatory quarantines to keep infected people in their homes).
Putting these plans into action will require considerable coordination among government officials, the media, and health-care workers—and widespread buy-in from the public.
Yet little discussion has appeared in the press to help people understand the measures they can take to best protect themselves during a flu outbreak—other than vaccination and antivirals.
Washing hands diligently, avoiding public places during an outbreak, and having a supply of canned goods and water on hand are sound defenses, he says.
Such steps could be highly effective in
helping to slow the spread of the virus. In Mexico, for instance,
where the first swine flu cases were identified in March, the
government launched an aggressive program to get people to wash
their hands and exhorted those who were sick to stay home and
effectively quarantine themselves. In the United Kingdom, the
national health department is promoting a “buddy” program,
encouraging citizens to find a friend or neighbor willing to deliver
food and medicine so people who fall ill can stay home.
Likewise, our encouragement of early treatment with antiviral drugs will likely lead many people to show up at the hospital at first sniffle.
All of which leaves open the question of what people should do when faced with a decision about whether to get themselves and their families vaccinated.
There is little immediate danger from getting a seasonal flu shot, aside from a sore arm and mild flu-like symptoms. The safety of the swine flu vaccine remains to be seen. In the absence of better evidence, vaccines and antivirals must be viewed as only partial and uncertain defenses against the flu. And they may be mere talismans.
By being afraid to do the proper studies now, we
may be condemning ourselves to using treatments based on illusion
and faith rather than sound science. |