|
Aberdeen, Scotland, UK
January 01, 2002
Independent studies are even fewer, moreover, no peer-reviewed publications exist in which the results of clinical investigations on the possible effects of GM food on human health are described.
Even though the evaluation of the safety or possible toxicity of GM foodstuffs is more difficult than that of drugs or food additives, this scarcity of data and the lack of a scientific database is curious particularly as descriptions of the results of chemical, nutritional and biological testing in some early (unpublished) studies or some more recent publications demonstrate the feasibility of carrying out proper and scientifically valid health risk assessment on GM foods.
In this review, after critically examining some of the basic principles, past results and possible novel methods of future health safety assessment of GM foodstuffs, the conclusion appears to be that as the tools for the recognition and indeed for the elimination of the risks GM foods may present for us are available or can be developed, it is the will and the funding for such work that needs to be found.
Lectins are thus regarded as natural insecticidal proteins. Based on this recognition, by transferring insecticidal lectin genes using recombinant gene methodologies, many novel genetically modified (GM) agronomically and commercially important crop plants have recently been developed which are resistant to their most devastating pests.
Currently one of the best known examples of insecticidal lectin genes from higher plants is the snowdrop (Galanthus nivalis) bulb lectin, GNA. From bacteria the lectin-endotoxins of Bacillus thuringiensis are the most widely used protective agents in GM plants. However, as the protective effect of the lectin is passive the plant tissues first need to be ingested before the released constitutive lectin could harm the pest/predator by interfering with nutrient assimilation in their digestive system.
It also needs to be recognized that if the same plant is eaten by
mammals this lectin may also react with their gut and harm their
health. Thus, there is an inextricable link between lectin-mediated
plant protection and potential nutritional and health problems for
the consumers.
Thus, as has been noted there are many opinions on the safety of GM food but very few data published in peer-reviewed science journals (Domingo, 2000).
This has recently been confirmed in a comprehensive scientific review (Pusztai, 2001). The almost total dependence of GM food regulation on the so-called principle of "substantial equivalence" and lack of independently verified safety testing is particularly regrettable.
The apparent similarity of macro/micro nutrient components in GM crops and their conventional counterparts established by chemical analysis, regardless of how sophisticated this may be, cannot reveal the presence of unknown toxic/allergenic components resulting from unintended effects of the insertion of a novel gene into the plant's genome.
Thus, GM food regulation is presently based on inadequate
and potentially dangerous principles.
Better chemical diagnostic methods are needed, including mRNA * fingerprinting, proteomics and secondary metabolite profiling (Kuiper et al., 2000).
* messenger Ribonucleic acid: The DNA code is transcribed within the cell nucleus into complementary mRNA, which in the cell protein synthesizing apparatus is then translated into the appropriate protein.
However, and most importantly, novel toxicological/nutritional methods need to be developed or present methods improved to be able to screen for the potential harmful consequences on human/animal health of GM food crops before these are allowed to enter the human food chain either directly or indirectly (Ewen & Pusztai, 1999a; Pusztai, 2000).
However, before
exploring the efficacy of possible novel biological testing methods
it may be instructive to review some studies which influenced the
development of testing.
# Calgene, Inc. is a US based biotechnology company that applied to the Food & Drug Administration (FDA) to sanction the release of the first commercial GM crop, the FLAVR-SAVRTM tomato developed by them.
This was despite the fact that the results of chemical analyses showed that there were no significant alterations in the composition of the tomatoes and therefore the GM and parent tomatoes were "substantially equivalent."
According to the FDA's evaluation the commissioned biological tests, in full agreement with the results of the chemical analyses, supported the safety of the GM tomatoes as no toxic effects were observed in the study. The weight gain, food consumption and clinical and haematology parameters of well-fed male/female rats which were also tube-fed with homogenized GM or conventional tomatoes, were claimed not to be statistically significantly different.
On the basis of all these tests the FDA
decided that FLAVR-SAVRTM tomatoes were as safe as conventional
tomatoes and, furthermore, that no toxicological testing of other GM
foodstuffs will in future be required.
However, as they are now on the internet (Alliance for Biointegrity, 1998) it was possible to subject the study to independent critical scrutiny. From this it is difficult to understand how the FDA came to the conclusion that these GM tomatoes were safe and needed no further studies. The fact that the tomatoes used in the different experiments were from different locations and harvested at different times should have at least put a serious question mark to the validity of the compositional comparisons in these studies and to the substantial equivalence of the GM and non-GM tomatoes (Pusztai, 2001).
Furthermore, the acute toxicity experiments were poorly designed and some of the methods used in the study were flawed.
Thus, in a major nutritional study of such importance more attention should have been given to selecting groups of rats whose starting weights were more closely matched. When differences in rat weights are close to twofold, as in these experiments, the large standard deviations in the data make it difficult to draw valid conclusions whether the weight gain, diet consumption, organ weights and other parameters of rats given GM or non-GM tomatoes were similar or different.
Even more importantly, no histology was done on the intestines although some of the female rats developed erosive/necrotic stomach lesions (Table 1).
Table 1
However, these were dismissed by the FDA because the lesions were claimed to be spontaneous/transitory and unrelated to GM tomatoes but probably due to mucolytic agents, food restriction and/or stress resulting from animal restraint.
However, tomatoes are not know to contain mucolytic agents, feed was provided ad lib, rats were not restrained or stressed and all test and control rats were treated the same way. As they were only tested once at the end of the experiment, it is difficult to maintain that the lesions were spontaneous or transitory. It also needs to be stressed that in these tests the rats were used as models for humans.
However, in human pathology it is not legitimate to use the term "mild erosion/necrosis" because glandular stomach erosion can lead to life-threatening haemorrhage, particularly in the elderly and in patients on non-steroidal anti-inflammatory agents. In the FDA study there was also an unexplained mortality of rats, apparently seven out of forty rats given GM tomatoes died within two weeks of the experiment.
The conclusion appears to be inescapable that as the whole FLAVR-SAVRTM study was poorly designed and executed and, most importantly, led to flawed conclusions, the claim that these GM tomatoes were as safe as conventional ones is at best premature and, at worst, faulty.
It was claimed that the growth, feeding value, the histopathology of immune related organs and the amounts of soybean-specific immunoglobulins showed no significant differences between animals fed GM and non-GM lines and no immunotoxic activity was found in GM soybean-fed rats or mice.
Unfortunately, although the design of this long-term study was acceptable its execution was poor. Thus, the growth of rats was unacceptably low and only amounted to just over 20 g over 105 days and the growth of mice was zero.
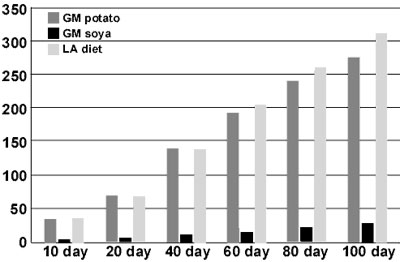
For comparison in the much-criticised GM potato study (temporarily displayed on the Rowett research Institute website in 1999 against my expressed wishes) in which the dietary protein concentration was only 8%, the rats grew close to 300 g during the same period of 105 days (Figure 1).
Figure 1
Growth rates of rats
(g/day) fed GM potato or GM soya diets for 105 days The GM potato experiments were published on the Rowett website (Rowett Research Institute, 1999) and the feeding studies with GM soya were by Teshima et al, 2000. For comparison, the growth rate of control rats fed on a high-quality diet containing lactalbumin (LA) are also included.
Thus, these unphysiological, basically, starvation conditions may have invalidated all the results.
In fact, this study gave a good example of how under starvation conditions most physiological/metabolic/immunological parameters could become unreliable, as demonstrated by the large variability and standard deviation of the mean values obtained for soybean-specific IgE and IgG in both rat and mice sera (Table 2).
Table 2
Despite these shortcomings, this study made a significant contribution to an eventual risk assessment protocol for GM foodcrops that could be generally accepted.
...FOR COMPOSITIONAL AND NUTRITIONAL/TOXICOLOGICAL TESTING OF GM FOOD
Selection of suitable genes
Obviously, only those genes can be accepted whose products have no apparent toxic/antinutritive effects in these biological tests. However, the lack of toxicity of this gene product as expressed in the transgenic plant must also be established. Thus, the gene product will have to be isolated from the transgenic plant and its biological, hormonal and immunological properties and allergenicity tested.
The present practice of using an E. coli recombinant gene product for the testing is not acceptable because post-translational modifications, which are different in prokaryotes and eukaryotes, can have major effects on the biological properties of the gene product proteins.
For this, in addition to quantitatively measuring the macronutrient (protein, DNA/RNA, carbohydrate, fat, etc) composition of the GM foodcrops and their parent lines, novel methods need to be developed to pinpoint any differences in minor constituents between GM and their parent lines (Kuiper et al., 1999).
However, for a rigorous and meaningful compositional comparison, the GM plant must be grown together with its non-GM parent line under identical and favourable conditions. For example, when this was done with GM and parent line potatoes significant differences were observed in a number of tuber constituents of nutritional importance (Rowett Research Institute, 1999).
Moreover, the differences were not only significant between the parent and GM lines but also between the two GM lines developed using the same vector construct and at the same time (Table 3).
It may also be prudent to grow the same GM and non-GM plants under non-ideal conditions because it is possible that compositional changes will occur in stressed plants and the GM and non-GM lines could be differently affected.
Table 3
Thus, "substantial equivalence" must be established under a variety of conditions and when it is claimed that the GM and non-GM crops are equivalent, the conditions of the comparison must be declared.
Thus, toxicity is an unhelpful concept and would also be difficult to assay at the low doses of the GM products found in GM food. In contrast, when nutritional studies are carried out in which diets containing the GM crop at high and variable concentrations are fed first to young rapidly growing laboratory animals in comparison with controls, these should reveal the possible harmful effects of the GM crop on animal metabolism, the development of their organs and immune and endocrine functions and gut flora.
As the normality of these together determine the
development of the young animal into healthy adults, this also may
guarantee the short-term safety of the GM crop.
Thus, all diets must be iso-proteinic and iso-energetic and fully supplemented with vitamins and essential minerals. The composition of the control diet containing the parent line should be as close to the GM diet as possible. In a second control diet, the parent line should be supplemented with the gene product isolated from the GM crop whose concentration should be the same as in the GM crop.
The GM crop should be fed both raw and after
heat-treatment.
The animals should be weighed daily and any abnormalities observed. Blood samples should be taken before, during and at the end of the feeding experiments for immune studies (immune responsiveness assays, Elispot, etc), hormone assays (insulin, CCK, etc), and determination of blood constituents.
At the end of the experiments the animals are killed, dissected, their guts rinsed and the contents saved for further studies (enzyme contents, GM products, DNA, etc), gut sections taken for histology, the wet and dry (after freeze-drying) weights of organs recorded, and the organs subjected to compositional analyses.
All these data can comprehensively characterize the health and
metabolic status of the animals and the behavior of the GM fed
animals can be directly compared with that of the controls.
Table 4
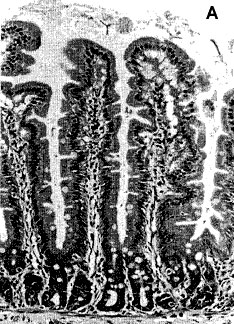
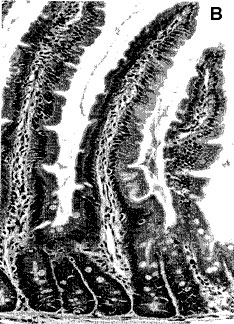
Thus, a follow up gut histology study (Ewen & Pusztai, 1999b, Figure 2) indicated the possibility that, due to the trophic effect of an unknown growth/proliferative signal of the GM potatoes, the small intestine of the GM potato-fed rats underwent hyperplastic growth.
Figure 2
Histology of jejunal sections of rats on GM potatoes raw GM potato diets for 10 days (b) in comparison with that of rats given parental line potato diets (a). The villus length is similar in both but intraepithelial lymphocyte cell counts appear to be increased on GM potato diet. (14 mm bar = 100 _m).
This was of particular significance because the jejunum was not enlarged when the parent line diet was supplemented with the gene product.
This was in line with previous observations which showed that GNA, the gene product, had negligible growth factor effect on the jejunum even when included in the diet at several hundredfold concentration compared with that expressed in the GM potato lines (Pusztai et al., 1990).
In a different study ileal sectrions of mice fed GM potatoes expressing Bacillus thuringiensis var. kurstaki Cry 1 toxin gene or the toxin itself similar hypertrophic and other changes in gut ultrasturcture were shown (Fares & El-Sayed, 1998), suggesting that GM potatoes may have common trophic effects on the gut.
Organ
weight changes could therefore be useful indicators of metabolic
events after feeding with diets containing GM foodstuffs,
particularly if followed up by histological examinations in the
safety assessment of GM crops.
Table 5
Although in the GM potato study no hormone assays were performed on rats fed GM or non-GM diets the consistent strong pancreatic growth stimulated by the GM potato diets in the feeding studies suggests that this possibly was the result of the release of CCK (cholecystokinin) or some other humoral growth factor from the duodenum by an unknown growth/proliferative signal only found in the GM potato diets.
Again, GNA could not be responsible for this because GNA does not stimulate the enlargement of the pancreas (Pusztai et al., 1990).
It would also be a good indicator to measure circulating insulin levels after ingestion of GM diets, particularly as insulin assays can easily be done on humans.
Accordingly, the GM crop cannot be accepted for inclusion in the
human/animal diet unless the differences were minor and the benefits
deemed to outweigh the potential harmful effects of the GM
foodstuff. However, such favorable risk/benefit analysis must be
confirmed by further independent research with animals closer to
humans and any final decision should be made by consensus openly,
transparently and inclusively and not secretly by companies which
have developed the GM crop.
Accordingly, this method of gene transfer and the resulting GM crop is unacceptable.
Thus, further research I needed into other, more precise and safer methods of genetic modification.
Since there were indications that the GM potatoes affected some of the male secondary sex organs in rats, similar studies also ought to be routinely performed with female small animals and extended to studies into the effects of GM foodstuffs on reproductive performance.
Furthermore, as some of the
potential effects of the consumption of GM foods is likely to be
manifested in the long-term, these reproductive studies should be
coupled to nutritional/toxicological tests in which offsprings of
successive generations brought up on GM food should be tested in
comparison with those reared on comparable non-GM diets.
Unlike the present methodology where the effects of the various constructs, promoters, plasmids, marker genes, etc on the mammalian gastrointestinal tract have never been properly investigated, in future recombinant gene technologies every step should be thoroughly tested before the development of novel GM crops.
However, even these will have to be subjected to rigorous animal testing as outlined above and then followed by drug-type certification tests before allowing them into the food chain. Some may think that the studies envisaged in this protocol are excessive but when it comes to the health and long-term prospects of the human race, nothing can be excessive. Moreover, the methods for staring up testing are available and many scientists are ready.
It is only the political will and the money which needs to be found.
|